A mole of any atoms has a mass in grams equal to the atomic weight of the. However, as seen by the helium and sulfur examples, the masses of individual atoms are not whole numbers. Individual carbon atoms therefore have a mass of either 12.000 or 13.03354 amu. By definition, the mass of the carbon-12 nucleus is 12u, where u is the atomic mass unit (amu), equal to 1.66 x 10-27 kg. No single carbon atom has a mass of 12.01 amu, but in a handful of C atoms the average mass of the carbon atoms is 12.01 amu. This is the average atomic mass of carbon. This is because most naturally occurring elements (including carbon) have more than one isotope. On the periodic table the mass of carbon is reported as 12.01 amu. The entire science of organic chemistry, which is the study of carbon compounds, is based on carbon.\). For example, atomic mass of carbon is not 12 u but 12.01 u. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. No form of life on the planet earth is without carbon in its structure. Every plant, animal, and everything in between that is alive is based on carbon. The most important impact carbon has on the human race is the fact that carbon is the basic building block of life, as we know it. Carbon-12 is composed of 6 protons, 6 neutrons, and 6 electrons. Other atoms typically do not have round-number atomic masses. Carbon-12, an atom of carbon-containing six neutrons, has an atomic mass of 12 amu. For example, carbons molar mass is 12.011 g/mol. Each element has its own unique molar mass. A single atom’s atomic mass is just its total mass, and it is commonly given in atomic mass units or amu. The mass in grams of one mole of substance is called molar mass. As an example, solid carbon is used to reduce iron from its oxide (Fe+3 to Fe0) in the blast furnace or other similar processes in which metallic elements are reduced from their oxide ores. Carbon-12 is of particular importance in its use as the standard from which atomic masses of all nuclides are measured, thus, its atomic mass is exactly 12 daltons by definition. The atomic mass of an element is the average mass of the atoms of an element. The atomic mass of an atom is a feature that is closely related to its mass number. The value of the mole is equal to the number of atoms in exactly 12 grams of pure carbon-12. Its ability to combine with oxygen makes it both a powerful and useful reducing agent (a substance that donates electrons resulting in reduction of the charge of the ion or atom being reduced). Carbon has a particularly strong affinity for oxygen either in the form of gaseous oxygen, or as oxygen contained in chemical combination with other elements. Pure carbon is a relatively reactive element and combines directly with many chemical elements, especially those considered oxidizing agents. Depending on the amount of graphite, amorphous carbon, or other contaminating elements, diamond can be found in colors ranging from clear water white through shades of black, gray, yellow, red, orange, blue, and green. The Avogadro constant has a unit: L 6.022 141 79 30 10 23 m o l 1. 
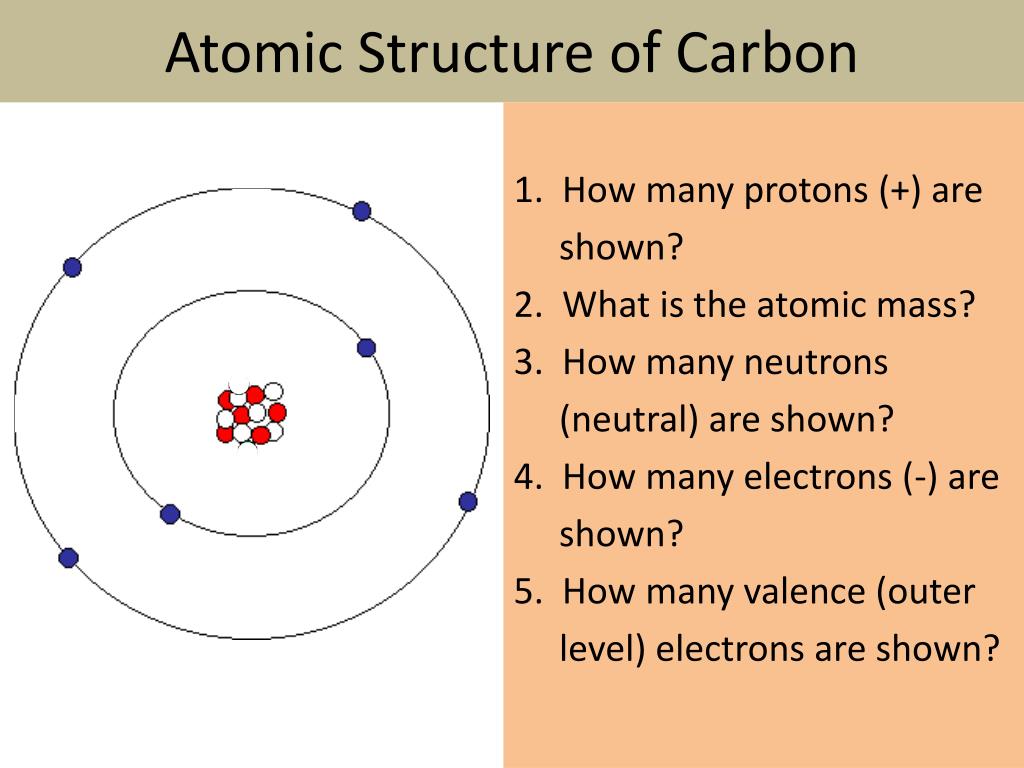
The unified atomic mass unit (u) is 1.660538921 × 10 24 grams. Other atoms dont generally have round-number atomic masses for. Most forms of carbon, excluding diamond, are black to grey-black in color. Avogadro's number is 6.02214129 × 10 23 and represents the number of carbon-12 atoms in 12 grams of unbound carbon-12 in the ground electronic state. By definition, an atom of carbon with six neutrons, carbon-12, has an atomic mass of 12 amu. Carbon occurs naturally in three isotopes: carbon 12, which has 6 neutrons (plus 6 protons equals 12). Some of these forms include hexagonal graphite, rhombohedral graphite, diamond, buckminsterfullerene, and amorphous carbon (not really a crystalline form). For example, carbon has six protons and is atomic number 6. 
It is found in the gas state at room temperature, and as the source of available carbon in the carbon cycle, atmospheric CO 2 is the primary carbon source for life on Earth. It is made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. There are a number of forms of carbon, known as allotropes, which are composed of pure carbon atoms but are arranged in different crystal lattices. Carbon dioxide is a chemical compound with the chemical formula CO2. Carbon is one of the few elements that occur in nature in its native or free elemental form.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
